Open Journal of Clinical and
Medical Images
Research Article - Open Access, Volume 5
Comparison of the efficacy of ultrasound-guided coil combined with glue injection and endoscopic glue injection in the treatment of gastric variceal bleeding: A meta-analysis
Hong-Bin Zhu; Yi-Fu Xia; Li-Xia Xin; Chun-Qing Zhang*
Department of Gastroenterology, Provincial Hospital of Shandong First Medical University, China.
*Corresponding Author: Chun-Qing Zhang
Department of Gastroenterology, Provincial Hospital of Shandong First Medical University, Jinan, Shandong 250021, China.
Email: zhangchunqing_sdu@163.com
Received : Jun 10, 2025
Accepted : Jul 22, 2025
Published : Jul 29, 2025
Archived : www.jclinmedimages.org
Copyright : © Chun-Qing Z (2025).
Abstract
Background: Endoscopic Cyanoacrylate Injection (E-CYA) is effective for Gastric Varices (GV) but carries risks like rebleeding and embolization. Endoscopic ultrasound-guided coil with glue-coating (EUS-GC) may reduce adverse events, but direct comparative efficacy/safety data was lacking.
Methods: This PRISMA-compliant meta-analysis (PROSPERO CRD42024496495) systematically searched databases (1995-2024) for studies directly comparing EUS-GC vs E-CYA for GV. Metaanalysis used RevMan 5.4.
Results: Six studies (413 patients: EUS-GC=168, E-CYA=245) were included. EUS-GC significantly reduced rates of posttreatment rebleeding (OR=0.23, 95% CI: 0.12~0.44, P<0.01), tissue adhesive dosage (SMD=-1.48, 95% CI: -2.30~-0.66, P<0.01), and need for reintervention (OR=0.18, 95% CI: 0.09~0.36, P<0.01) compared to E-CYA. However, there were no statistically significant differences between the groups in rates of postoperative adverse events (OR=0.70, 95% CI: 0.26~1.89), embolization in the systemic circulation (OR=0.33, 95% CI: 0.11~0.98), or all-cause mortality (OR=0.42, 95% CI: 0.10~1.67).
Conclusion: This meta-analysis demonstrates that EUSGC offers significant advantages over E-CYA in reducing posttreatment rebleeding, glue usage, and the need for reintervention in GV patients. While showing favorable trends, EUS-GC did not significantly differ from E-CYA regarding overall postoperative adverse events, systemic embolization, or mortality.
Keywords: Liver cirrhosis; Ultrasound endoscopy; Cyanoacrylate; Gastric varices; Meta-analysis.
Citation: Hong-Bin Z, Yi-Fu X, Li-Xia X, Chun-Qing Z. Comparison of the efficacy of ultrasound-guided coil combined with glue injection and endoscopic glue injection in the treatment of gastric variceal bleeding: A meta-analysis. Open J Clin Med Images. 2025; 5(2): 1205.
Introduction
Rupture and hemorrhage of esophagogastric varices is one of the major complications of portal hypertension. Although esophageal varices account for the vast majority of gastrointestinal bleeding cases, Gastric Varices (GV) only account for 20% of such cases. However, compared with esophageal varices, GV are associated with a higher risk of uncontrollable bleeding, a more urgent need for blood transfusion, and higher rates of postoperative rebleeding and mortality [1,2]. The cumulative risk of bleeding within 5 years can reach up to 44% [3]. Therefore, implementing rational therapeutic interventions can significantly reduce the mortality rate of patients with portal hypertension.
Based on the Sarin classification, esophagogastric varices are categorized into four distinct types: type 1 Gastroesophageal Varices (GOV1), which extend along the pylorus and the lesser curvature; type 2 Gastroesophageal Varices (GOV2), characterized by their extension to the fundus of the stomach; Isolated Gastric Varices type 1 (IGV1), confined solely to the gastric fundus; and Isolated Gastric Varices type 2 (IGV2), which can occur in any part of the stomach [4].
GV can be managed through various therapeutic approaches, including the use of non-selective beta-Blockers (NSBBs), balloon tamponade, Transjugular Intrahepatic Portosystemic Dhunt (TIPS), and Balloon-occluded Retrograde Transvenous Obliteration (BRTO) [5]. For patients with GOV2, IGV1, and IGV2, Endoscopic Cyanoacrylate injection (E-CYA) using tissue adhesives such as n-butyl-2-cyanoacrylate currently represents the optimal therapeutic approach, demonstrating treatment success rates ranging from 58% to 100% [6]. However, this therapeutic approach is associated with a spectrum of severe complications, predominantly thrombotic events [7].
Endoscopic Ultrasound-Guided Glue Combined with coil embolization (EUS-GC) represents an innovative therapeutic modality for GV, initially introduced by Binmoeller et al., which has demonstrated a remarkable variceal occlusion rate of up to 96% [8]. Literature analysis has confirmed that EUS-GC is both safe and effective for patients with GV, warranting its consideration in clinical practice [9]. However, to date, no meta-analysis has been conducted to determine whether EUS-GC confers superior therapeutic efficacy compared to E-CYA for the treatment of GV. The present meta-analysis aims to systematically review existing literature and compare the clinical outcomes of these two treatment modalities in managing GV.
Materials & methods
Search methodology
We systematically searched Medline, the Cochrane Library, Embase, Web of Science, and other relevant electronic databases for published articles and reports that compared EUS-GC with E-CYA in the treatment of GV, with the publication time limited from January 1995 to July 2024, using the search terms “endoscopic ultrasound”, “coil”, “glue”, “cyanoacrylate”, and “gastric varices”, and the specific search strategy was (“Gastric Varices” OR “Gastric Varix”) AND (Hemorrhages OR Hemorrhage OR Bleeding) AND (endoscopy OR EUS OR endoscopic ultrasound) AND (glue OR “tissue adhesive” OR cyanoacrylate OR enbucrilate OR bucrilate OR butylcyanoacrylate OR “butyl cyanoacrylate” OR coil OR “combination therapy”). After retrieving the relevant literatures, we first removed duplicates from the screened literatures to exclude possible duplicate or overlapping data, then read the titles and abstracts of all qualified literatures to conduct a preliminary summary and review, and finally determined the literatures that met the research criteria through a close reading of the full texts.
Inclusion criteria
(1) Randomized controlled trials or retrospective studies comparing EUS-GC with E-CYA in the treatment of GV, with language restricted to English.
(2) Patients with cirrhotic portal hypertension complicated by GV, regardless of disease duration or etiology.
(3) Articles that include a comparison of the therapeutic efficacy between EUS-GC and E-CYA.
Exclusion criteria
(1) Studies reporting outcomes of EUS-GC or E-CYA only, without comparative analysis.
(2) Duplicate publications.
(3) Reviews, case reports, single-arm studies, and meta-analyses.
(4) Studies with no full-text available or lack extractable data.
Data extraction and quality assessment
The relevant data of each study were independently extracted into a standardized form by two authors (Hongbin Zhu and Lixia Xin), and then independently verified by another two authors (Chunqing Zhang and Yifu Xia) to ensure data accuracy. If necessary, the corresponding authors of the original studies would be contacted via email to obtain additional data or clarify uncertainties. The quality of included studies was independently assessed by the same two authors (Hongbin Zhu and Lixia Xin) using both the Newcastle-Ottawa Scale and the Cochrane Collaboration’s risk-of-bias tool.
Outcome definitions
The primary outcome was the post-treatment rebleeding rate. Secondary outcomes included the rate of reintervention, overall adverse event rate, systemic embolism rate, and postoperative mortality.
Statistical analysis
The statistical analysis for this meta-analysis was conducted using Review Manager 5.4 (RevMan 5.4). Heterogeneity among the included studies was assessed using both the I² statistic and Q test, with heterogeneity considered low when P>0.1 and I²<50%, in which case a fixed-effect model was applied; otherwise, a random-effects model was used, while a random-effects model was prioritized after excluding significant clinical and methodological heterogeneity. Dichotomous variables were analyzed using Odds Ratios (OR) and continuous variables were analyzed using Standardized Mean Differences (SMD), with 95% Confidence Intervals (CI) calculated for all variables and statistical significance set at P<0.05.
Results
Search results
The initial search identified 2690 potentially relevant studies. After removing duplicates and excluding studies unrelated to the research topic, 337 articles remained for further evaluation. Following the exclusion of studies reporting only EUS-GC or only E-CYA outcomes, a final set of 6 studies met all inclusion criteria and were included in the meta-analysis [10-15]. A total of 413 patients were included in the analysis. All selected studies provided direct comparative data on the therapeutic efficacy of both treatment modalities for GV.
Detailed aggregated data including age, gender, GV classification, number of treatment sessions, volume of tissue adhesive used, number of coils deployed, as well as treatment outcomes and adverse events are comprehensively presented in Table 1.
Table 1: Basic characteristics and quality evaluation of 6 included literature.
| First author | Design | Groups | Number (n) | M/F (n) | Age (years) | Classification | Follow-up time | Quality |
|---|---|---|---|---|---|---|---|---|
| Maíra Ribeiro de Almeida LÔBO [15] | RCT, Brazil | EUS-GC | 16 | 8/8 | 49.31±14.83 | GOV2/IGV1 13/3 | 9.94+/-6.76 (M) | high |
| E-CYA | 16 | 5/11 | 57.69±11.56 | GOV2/IGV1 13/3 | 9.94+/-6.76 (M) | |||
| Carlos Robles-Medranda [14] | Retrospective, Ecuador | EUS-GC | 17 | 10/7 | 63.29±8.8 | GOV2/IGV1 12/5 | Medium | |
| E-CYA | 19 | 10/9 | 62.83±11.5 | GOV2/IGV1 12/7 | ||||
| T. S. A. Satya Maharshi [13] | retrospective | EUS-GC | 16 | |||||
| E-CYA | 16 | |||||||
| Kapil D. Jamwal [12] | Retrospective, India | EUS-GC | 40 | 30/10 | 44.1±8.23 | 201 (94–378) (D) | high | |
| E-CYA | 40 | 28/12 | 43.2±7.88 | 193 (75–301) (D) | ||||
| Jayanta Samanta [11] Retrospective, India and Italy | EUS-GC | 58 | 42/16 | 44.33±12.1 | GOV2/IGV1 31/27 | 701.5 (D) | high | |
| E-CYA | 118 | 83/35 | 46.63±13.6 | NA | 701.5 (D) | |||
| DW Chen [10] | Retrospective, China | EUS-GC | 21 | 14/7 | 57.5±7.7 | GOV1/GOV2/IGV1 1/13/7 | 13.0 (8–16) (M) | high |
| E-CYA | 36 | 27/9 | 58.8±8.0 | GOV1/GOV2/IGV1 2/19/15 | 12.5 (8–15.75) (M) |
Meta-analysis results
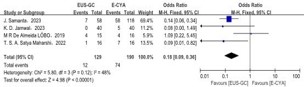
Postprocedural rebleeding: All six included studies compared post-treatment rebleeding rates between the treatment groups, with 168 patients in the EUS-GC group and 245 patients in the E-CYA group. Analysis using a fixed-effects model showed that the post-procedure bleeding rate was significantly lower with EUS-GC treatment compared to E-CYA treatment (OR=0.23, 95% CI: 0.12-0.44, P<0.05) (Figure 1a). Considering that most patients underwent endoscopic therapy for either active bleeding or prior bleeding history, we performed subgroup analyses based on bleeding status. The data analysis demonstrated that patients with active bleeding or prior bleeding history who received EUS-GC treatment also had lower rebleeding rates than those treated with E-CYA (OR=0.10, 95% CI: 0.04-0.27, P<0.05) (Figure 1b).
Volume of tissue adhesive: Three studies reported and compared the volume of tissue adhesive used during treatment, including 77 patients in the EUS-GC group and 92 patients in the E-CYA group. Due to significant heterogeneity among studies (P<0.1, I2=81%), a random-effects model was applied for analysis. The results demonstrated a significant difference in tissue adhesive volume between EUS-GC and E-CYA (SMD=-1.48, 95% CI: -2.30 to -0.66, P<0.05) (Figure 2), with lower tissue adhesive usage observed in the EUS-GC group.
Need for retreatment: Four studies compared the rate of requiring repeat embolization therapy after treatment, involving 129 patients in the EUS-GC group and 190 patients in the E-CYA group. With low heterogeneity among studies (P>0.1, I2=48%), a fixed-effects model was applied for analysis. The results demonstrated a significantly lower proportion of patients requiring repeat embolization therapy in the EUS-GC group compared to the E-CYA group (OR=0.18, 95% CI: 0.09–0.36, P<0.05) (Figure 3).
Post-treatment adverse events
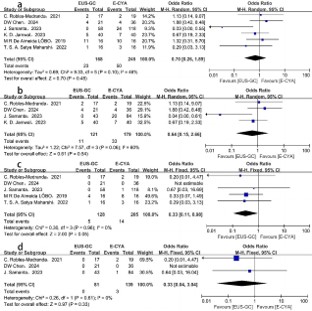
All six included studies reported the incidence of post-treatment adverse events, involving 168 patients in the EUS-GC group and 245 patients in the E-CYA group. Due to moderate heterogeneity among studies (P=0.1, I2=46%), a random-effects model was employed for analysis. The results showed no statistically significant difference in overall adverse event rates between the EUS-GC and E-CYA groups (OR=0.70, 95% CI: 0.26– 1.89, P>0.05) (Figure 4a). Subgroup analysis of patients with active bleeding or prior bleeding history similarly demonstrated no significant difference in adverse event rates (OR=0.64, 95% CI: 0.15–2.66, P>0.05) (Figure 4b). Given that systemic embolism represents one of the most severe postprocedural complications for both therapies, a separate analysis was performed.
The results indicated no statistically significant difference in systemic embolism rates between the two treatment modalities (OR=0.33, 95% CI: 0.11–0.98, P=0.05) (Figure 4c). Similarly, among patients with active bleeding or prior bleeding history, the incidence of systemic embolism showed no significant difference (OR=0.33, 95% CI: 0.04–3.04, P>0.05) (Figure 4d).
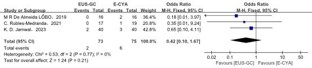
Post-treatment mortality: Three studies compared all-cause mortality after treatment, including 73 patients in the EUS-GC group and 75 patients in the E-CYA group. With no observed heterogeneity among studies (P>0.1, I2=0%), a fixed-effects model was applied for analysis. The results demonstrated no statistically significant difference in all-cause mortality between the EUS-GC and E-CYA groups (OR=0.42, 95% CI: 0.10–1.67, P>0.05) (Supplemental Figure 1).
Publication bias
Due to the limited number of included studies, formal assessment of publication bias was not performed in this metaanalysis.
Discussion
Approximately 20% of patients with cirrhosis and portal hypertension develop GV [2]. Although GV hemorrhage carries a lower baseline bleeding risk compared to esophageal varices, acute GV bleeding events are typically more severe, with higher risks of rebleeding and mortality [16]. Endoscopic management of GV by subtype: GOV1, as extensions of esophageal varices, can be effectively managed with conventional endoscopic band ligation. GOV2/IGV1/IGV2 subtypes are more appropriately treated with tissue adhesive injection cyanoacrylate to optimize hemorrhage control and reduce recurrence risk [17], an approach endorsed by the Baveno VII consensus for secondary prevention of GV bleeding [18]. While endoscopic cyanoacrylate injection achieves excellent immediate hemostasis, its clinical utility is limited by a postprocedural rebleeding rate of up to 36% [19], potentially attributable to incomplete variceal obliteration. Furthermore, the traditional method of assessing the adequacy of obliteration by inspecting coagulum formation in the injected varices remains subjective, potentially leading to misinterpretation and subsequent rebleeding risks [20]. Beyond rebleeding, systemic embolism caused by adhesive migration represents a critical safety concern, including cerebral, pulmonary, coronary, and splenic embolic events. Rare complications such as needle entrapment within varices, fistulation, and sepsis have also been documented [21]. EUS-GC therapy is emerging as a viable alternative to E-CYA [22], EUS helps endoscopists to clearly visualize the occlusion of varicose veins in real time and precisely target the veins that need to be punctured, thus minimizing the amount of tissue adhesive used while significantly reducing the risk associated with embolization [23].
Our study exhibits distinct methodological and analytical distinctions from previously published meta-analyses. The meta-analysis by Mohan et al [24] evaluated ultrasound-guided therapies versus E-CYA for GV management, with primary outcomes including rebleeding and variceal recurrence. Their results indicated that EUS-guided approaches achieved higher hemostasis rates than E-CYA, though comparable post-treatment rebleeding rates were observed between modalities. Notably, in subgroup analyses, EUS-GC demonstrated superior efficacy compared to other EUS-guided approaches. Bang et al [25] reported a preliminary abstract-form study comparing direct endoscopic cyanoacrylate injection with EUS-guided cyanoacrylate injection for GV. The authors documented higher rates of recurrent rebleeding events and overall adverse events in the direct endoscopic group versus the EUS-guided cohort. In contrast to previously published studies, this meta-analysis analyzes for the first time the pooled data of EUS-GC versus E-CYA in GV treatment separately.
This study demonstrates significant methodological strengths, including a systematic literature search with well-defined inclusion criteria, a rigorous screening process to exclude duplicate and low-quality studies, and detailed data extraction from high-quality evidence. All included studies were subjected to comprehensive quality assessment to ensure the robustness of findings. The analysis encompassed extensive objective endpoints, including post-treatment rebleeding rates, technical success rates, volume of tissue adhesive used, systemic embolism rates, and overall adverse event rates, thereby providing a comprehensive and scientifically rigorous evaluation of the comparative efficacy and safety profiles between EUS-GC and E-CYA for gastric varices management.
The limitations of this study reflect inherent constraints of meta-analytic methodology. First, the included studies were retrospective in design, which may introduce selection bias. Second, the absence of individual patient-level data precluded causal attribution for post-treatment bleeding events and mortality. Third, the technical proficiency required for both procedures is operator-dependent, potentially introducing performance variability that may compromise the generalizability of bleeding rates and adverse event outcomes across different clinical settings.
Conclusions
The data analysis of this study indicates that EUS-GC is a safe and effective treatment for GV. It demonstrated significant superiority over E-CYA in reducing post-treatment rebleeding rates, intraprocedural tissue adhesive volume, and need for repeat embolization. However, the advantages of EUS-GC in clinical efficacy rates, systemic embolism incidence, and overall post-treatment adverse events did not reach statistical significance. These findings warrant the initiation of large-scale, high-quality randomized controlled trials to further validate the potential benefits of EUS-GC and establish its broader clinical applicability.
Declarations
Funding: National Natural Science Foundation of China (81970533).
Conflict of interest statement: The authors have no conflicts of interest to declare.
References
- Ryan BM, Stockbrugger RW, Ryan JM. A pathophysiologic, gastroenterologic, and radiologic approach to the management of gastric varices. Gastroenterology. 2004; 126: 1175–89.
- Garcia-Tsao G, et al. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the Study of Liver Diseases. Hepatology. 2017; 65: 310–35.
- Kim T, et al. Risk factors for hemorrhage from gastric fundal varices. Hepatology. 1997; 25: 307–12.
- Sarin SK, Kumar A. Gastric varices: Profile, classification, and management. Am J Gastroenterol. 1989; 84: 1244–9.
- Tripathi D, et al. The role of the transjugular intrahepatic portosystemic stent shunt (TIPSS) in the management of bleeding gastric varices: Clinical and haemodynamic correlations. Gut. 2002; 51: 270–4.
- Hidaka H, Uojima H, Kokubu S. [Endoscopic treatments for portal hypertension]. Nihon Shokakibyo Gakkai Zasshi. 2019; 116: 395–403.
- Wani ZA, et al. Gastric varices: Classification, endoscopic and ultrasonographic management. J Res Med Sci. 2015; 20: 1200–7.
- Binmoeller KF, et al. EUS-guided transesophageal treatment of gastric fundal varices with combined coiling and cyanoacrylate glue injection. Gastrointest Endosc. 2011; 74: 1019–25.
- Baig M, Ramchandani M, Puli SR. Safety and efficacy of endoscopic ultrasound-guided combination therapy for treatment of gastric varices: A systematic review and meta-analysis. Clin J Gastroenterol. 2022; 15: 310–9.
- Chen D, Fu S, Shen R. Efficacy and safety of EUS-guided coil embolization in combination with cyanoacrylate injection versus conventional endoscopic cyanoacrylate injection in the treatment of gastric varices with spontaneous portosystemic shunts. Gastroenterol Rep (Oxf). 2024; 12: goae026.
- Samanta J, et al. EUS-guided coil and glue injection versus endoscopic glue injection for gastric varices: International multicentre propensity-matched analysis. Liver Int. 2023; 43: 1783–92.
- Jamwal KD, et al. Endoscopic ultrasound-guided coiling and glue is safe and superior to endoscopic glue injection in gastric varices with severe liver disease: A retrospective case control study. Clin Endosc. 2023; 56: 65–74.
- Satya Maharshi TSA, et al. Safety and efficacy of endoscopic conventional cyanoacrylate glue vs EUS-guided coil plus cyanoacrylate technique in the treatment of gastric varices: A randomized controlled trial (NCT05038319). United Eur Gastroenterol J. 2022; 10: 1037.
- Robles-Medranda C, et al. Cost-effectiveness of endoscopic ultrasound-guided coils plus cyanoacrylate injection compared to endoscopic cyanoacrylate injection in the management of gastric varices. World J Gastrointest Endosc. 2021; 13: 13–23.
- Lôbo MRA, et al. Safety and efficacy of EUS-guided coil plus cyanoacrylate versus conventional cyanoacrylate technique in the treatment of gastric varices: A randomized controlled trial. Arq Gastroenterol. 2019; 56: 99–105.
- Sarin SK, et al. Prevalence, classification and natural history of gastric varices: A long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992; 16: 1343–9.
- Vozzo C, et al. Management of gastric varices: GI perspective. Dig Dis Interv. 2023; 7: 266–72.
- de Franchis R, et al. Baveno VII Renewing consensus in portal hypertension. J Hepatol. 2022; 76: 959–74.
- Qiao W, et al. Cyanoacrylate injection versus band ligation in the endoscopic management of acute gastric variceal bleeding: Meta-analysis of randomized, controlled studies. Medicine (Baltimore). 2015; 94: e1725.
- Saraswat VA, Verma A. Gluing gastric varices in 2012: Lessons learnt over 25 years. J Clin Exp Hepatol. 2012; 2: 55–69.
- Seewald S, et al. A standardized injection technique and regimen ensures success and safety of N-butyl-2-cyanoacrylate injection for the treatment of gastric fundal varices. Gastrointest Endosc. 2008; 68: 447–54.
- McCarty TR, et al. Combination therapy versus monotherapy for EUS-guided management of gastric varices: A systematic review and meta-analysis. Endosc Ultrasound. 2020; 9: 6–15.
- Sabry F, et al. EUS-guided cyanoacrylate injection into the perforating vein versus direct endoscopic injection in the treatment of gastric varices. Endosc Int Open. 2023; 11: E202–10.
- Mohan BP, et al. Efficacy and safety of endoscopic ultrasoundguided therapy versus direct endoscopic glue injection therapy for gastric varices: Systematic review and meta-analysis. Endoscopy. 2020; 52: 259–67.
- Bang JY, et al. Comparison of direct endoscopic injection and EUS-guided fine needle injection of 2-octyl-cyanoacrylate for treatment of gastric varices. Gastrointest Endosc. 2015; 81: AB437.